Aptiv Releases Q1 2026 Financial Results
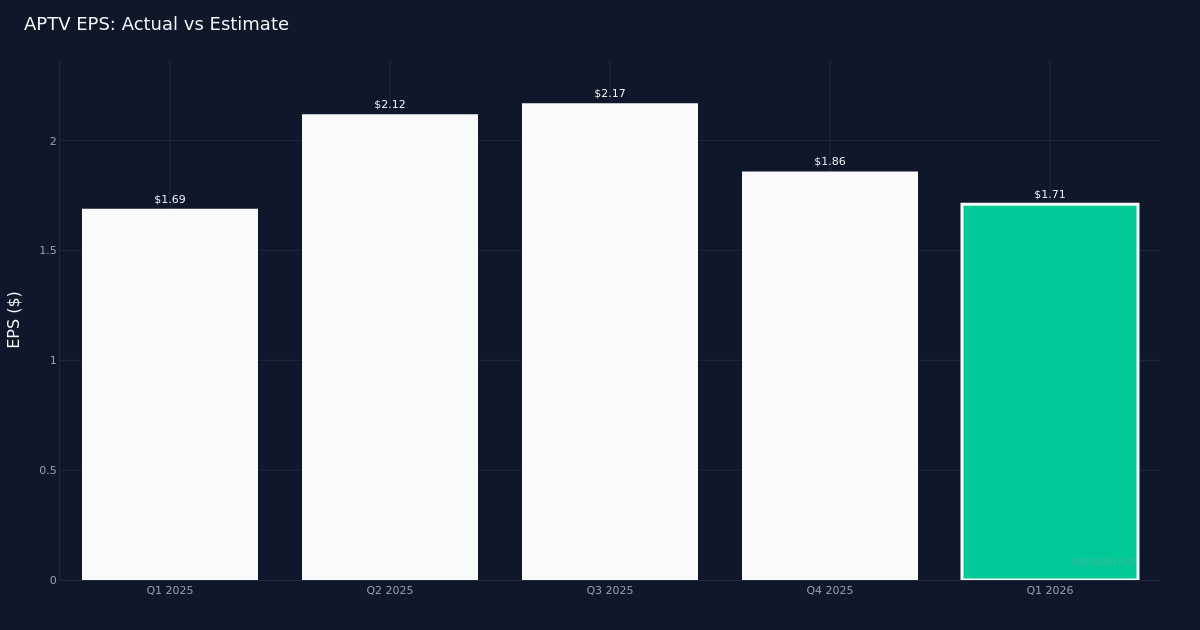
Aptiv PLC reported first-quarter 2026 results that exceeded Wall Street expectations, with adjusted earnings of $1.71 per share against an estimated $1.60. The automotive technology supplier generated $5.09 billion in revenue, a 5.0% increase year-over-year, driven primarily by strong performance in its Signal and Power Solutions segment. The company also provided adjusted earnings guidance for the full fiscal year 2026, ranging from $5.70 to $6.10 per share.
https://news.alphastreet.com/aptiv-releases-q1-2026-financial-results/